| Panel | ||||||||
|---|---|---|---|---|---|---|---|---|
|
|
|
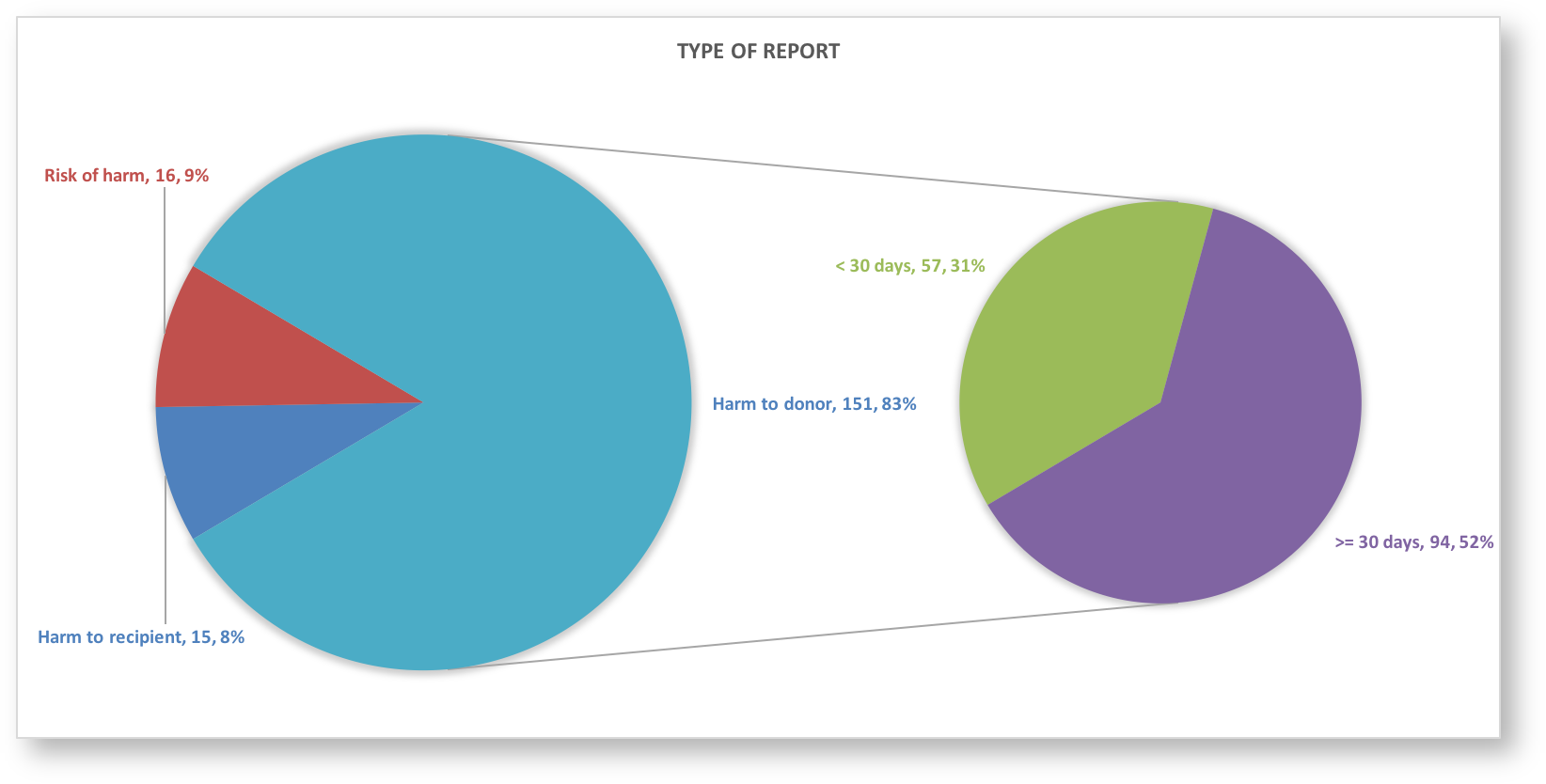
WMDA has set up a central global reporting system |
to report Serious (Product) Events and Adverse Reactions – S(P)EARs – to the WMDA. To access the reporting system, you login with your email address and password. If you do not have a password, contact the WMDA office. Aim
Purpose
|
|
donor
|
| Warning | ||
|---|---|---|
| ||
The SPEAR Committee reviews all reported incidents once a week. If you have a rapid alert that needs to be shared with the community quickly, we urge you to submit it via the online reporting tool and to immediately send an e-mail to sear-spear@wmda.info or call us on: +31 88 505 7900. |
Quick links
In case of recall or big risks for future donations or transfusions a report can be submitted as a rapid alert. This includes, but is not limited to:
|
| Info | ||
|---|---|---|
| ||
Quick links |
| Info | ||
|---|---|---|
|
Link to annual reports |
- WMDA S(P)EAR report 2018
- WMDA S(P)EAR report 2017
- WMDA S(P)EAR report 2016
- WMDA S(P)EAR report 2015
- WMDA S(P)EAR report 2014
- WMDA S(P)EAR report 2013
- WMDA S(P)EAR report 2012
- WMDA S(P)EAR report 2011
- WMDA S(P)EAR summary report 2003 - 2010
overview (for WMDA members only)
|
| Info | ||
|---|---|---|
|
SPEAR rapid alerts |
| Gallery | ||||||
|---|---|---|---|---|---|---|
|
Find below the link to the public WMDA webpage, where you will find all rapid alerts sent out.
|