CBB Survey 2015
These data are submitted by the cord blood bank to the WMDA in February 2015. The WMDA makes no representations or express or implied warranties regarding any information on this site or obtained through these links; expressly disclaims all legal liability and responsibility for the accuracy, completeness, currency, suitability, validity, or usefulness of such information; and is not and will not be liable for any statements, errors, or omissions in posted information, or for any losses, injuries, or damages that arise or are alleged to arise from such information. Use of any information provided on this site does not and is not intended to create a contractual or other relationship. All information is on an as-is basis. The data cannot be updated.
| 1. General Information | |
|---|---|
| Name of CBB | Bayerische Stammzellbank gGmbH |
| CBB Director | Hans |
| CBB Director | Knabe |
| Address | Robert-Koch-Allee 23 |
| Address | 82131 |
| Address | Gauting |
| Phone Number | ++49-89-89 32 66 220 |
| Website | www.akb.de |
| Date CBB Started Collecting Cord Blood Units (month/day/year) | 10/01/1998 |
| Number of Public Cord Blood Units | 4,021 |
| Planned Number of Public Cord Blood Units Stored in 2015 | 200 |
| Lists on BMDW | Yes |
| Affiliated with National Stem Cell Donor Registry | Yes |
| Registry Affiliation | ZKRD |
| 2. Cord Blood Units in Inventory | |
| Current Processing Method | Liquid+ red auto |
| Previous Processing Method | Liquid+ red manual |
| Year Current Process Method Started | 2010 |
| Percent of Units Plasma and RBC Reduced (manual) | 73 |
| Percent of Units Plasma and RBC Reduced (automated) | 27 |
| 3. Accreditations, Licenses and Certifications | |
| FACT-Netcord | Yes |
| AABB | No |
| Competent Authority/ National Health Authority | Yes |
| Name of Competent Authority | Regierung von Oberbayern and: Paul-Ehrlich-Institut |
| Audited by National Stem Cell Registry | Yes |
| ISO | No |
| Other | Marketing Authorization (Paul-Ehrlich-Institut), WMDA |
| 4. Cord Blood Collection | |
| Current Collection Practice Is the collection In/Ex -Utero or both? | In-utero |
| Collection Bag | Double needle |
| Agitation during Collection | Manual |
| 5. Conditioning/Transport from Collection Site to CBB | |
| Secondary Bag Used | Yes |
| Transport Conditions | Insulating transport container |
| Transport Conditions | Electronic temperature probe |
| Transport Conditions | Ground transport |
| Temp. for Storage and Transport | Other (lower limit +1-35°C, higher limit +6-30°C) |
| 6. Pre-Processing Evaluation | |
| Completed Prior to Accepting a CBU | Medical history, collection report, informed consent |
| Method for CD34 Remuneration | ISHAGE guidelines |
| External Proficiency Testing for QC of FACS Lab | Yes, other |
| External Proficiency Testing for QC of FACS Lab | INSTAND |
| Post Processing/ Pre Freeze CD34+ Cell Count | Yes |
| Time from Collection to Processing | up to 48H |
| 7. Processing and Packaging | |
| Pre Freeze Processing Methods- Unit in Inventory | SEPAX |
| Pre Freeze Processing Methods- Unit in Inventory | Volume reduction with HES-Manual |
| Pre Freeze Processing Methods- Current | SEPAX |
| Additives Currently in Use | HES |
| Current Cryopreservation Method | Conventional CRF |
| Current Cryopreservation Method | Other: Consarctic |
| Current Cryoprotectant Additive | Other: HES and autologous plasma |
| Current Cryobag | Single bag (one fraction) |
| Current Cryobag | Multiple bags |
| Current Target Cryopreservation Volume (mL) | 29.0 |
| Current Packaging for Storage | Canister |
| Current Packaging for Storage | More than one segment |
| 8. Testing | |
| PCR Performed on IgM+ Result | CMV |
| Extra Material Currently Stored | Cord blood DNA |
| Extra Material Currently Stored | Cord blood material for DNA extraction |
| Extra Material Currently Stored | Plasma/cord blood |
| Extra Material Currently Stored | Maternal material for DNA extraction |
| Extra Material Currently Stored | Maternal plasma/serum |
| Current Post Processing Threshold for Accepting a CBU for Public Use TNC | NA |
| Current Post Processing Threshold for Accepting a CBU for Public Use CD34 (10^6) Single Platform | 1.50 |
| Current Post Processing Threshold for Accepting a CBU for Public Use CD34 (10^6) Double Platform | NA |
| Current Post Processing Threshold for Accepting a CBU for Public Use CFU-GM | NA |
| Current Post Processing Threshold for Accepting a CBU for Public Use CFU | 1 |
| Current Post Processing Threshold for Accepting a CBU for Public Use Viability | 90 |
| 9. Storage | |
| Type of Storage Container Used | Conventional tank liquid phase |
| Type of Storage Container Used | Double walled liquid nitrogen |
| Monitoring of Storage | Centralized system remote monitoring |
| Monitoring of Storage | LN2 level |
| Monitoring of Storage | Lid opening |
| Monitoring of Storage | System default |
| Monitoring of Storage | Temperature monitoring |
| 10. HLA Typing | |
| Current Level of HLA Typing at Time of Listing HLA-A | HR |
| Current Level of HLA Typing at Time of Listing HLA-B | HR |
| Current Level of HLA Typing at Time of Listing HLA-C | HR |
| Current Level of HLA Typing at Time of Listing HLA-DRB1 | HR |
| Current Level of HLA Typing at Time of Listing HLA-DQB1 | HR |
| Current Level of HLA Typing at Time of Listing HLA-DPB1 | |
| Accreditation of HLA Lab | EFI accredited lab |
| Average Turnaround Time for Extended HLA Typing Results in days | 4 |
| Attached Segment Used for Confirmatory/ Verification Typing | No |
| Units Listed without Attached Segment and have not been Previously Typed on Attached Segment | No |
| Percentage of CBUs that have an Attached Segment | 90-100% |
| Confirmatory/ Verification Typing on an Attached Segment is Pre-Release Requirement | Yes |
| 11. Reservation and Cancellation Policies | |
| What Point is a CBU Reserved for a Patient | Reservation request |
| What Point is a CBU Reserved for a Patient | Shipment request |
| What Point is a CBU Reserved for a Patient | Other: At time of HLA typing request: CBU marked as requested for another patient |
| Length of Time a CBU can be Reserved in days | 90 |
| Reservation Fee | No |
| Reservation Cancellation Fee in Absence of Shipment Request | No |
| Can Reservation be Extended | Yes |
| Is a Unit Report Provided on a Unit that is Reserved for Another Patient | No |
| Is TC Informed when CBU is Released | Yes |
| 12. Release and Shipment | |
| Hemoglobinopathy Screening Performed Prior to Release | Yes |
| Criteria to Ship a CBU Viability and Cell Count | >60% / NA |
| Current Packaging for Shipment to TC | Metal canister |
| Current Packaging for Shipment to TC | 2 attached segments |
| Time Between Shipment Request and Sending CBU | Other |
| Time to Prepare a Cord Blood Unit for Shipment | Time dependent on extent of QC-testings to be completed prior to shipment (i.e. confirmatory typing). Without further testings: 0-3 days |
| Fee for Shipment Cancellation | Yes |
| Dry Shippers Validated to Maintain Temperature of at least -150 for 48 hours Beyond Expected Arrival | Yes |
| Electronic Temperature Data Logger on All Dry Shippers | Yes |
| Who Selects Transport Company | CBB |
| Who Selects Transport Company | accompagnied transport only (Ontime Courier or members of the fire department Munich); fee for transfer included in fee for CBU |
| Shape of Transport Container | Other |
| Shape of Transport Container | Onboard transport or transport by train: Little dry shipper in a cage of metal (cylindrical); transport by car: Dry shipper in outer container (cubic or mushroom) |
| 13. Adverse Events Reporting | |
| Who are S(P)EARS Reported To Competent Authority | Yes |
| Who are S(P)EARS Reported To Internal Report | Yes |
| Who are S(P)EARS Reported To National Registry | Yes |
| Who are S(P)EARS Reported To Transplant Center | Yes |
| Who are S(P)EARS Reported To WMDA | Yes |
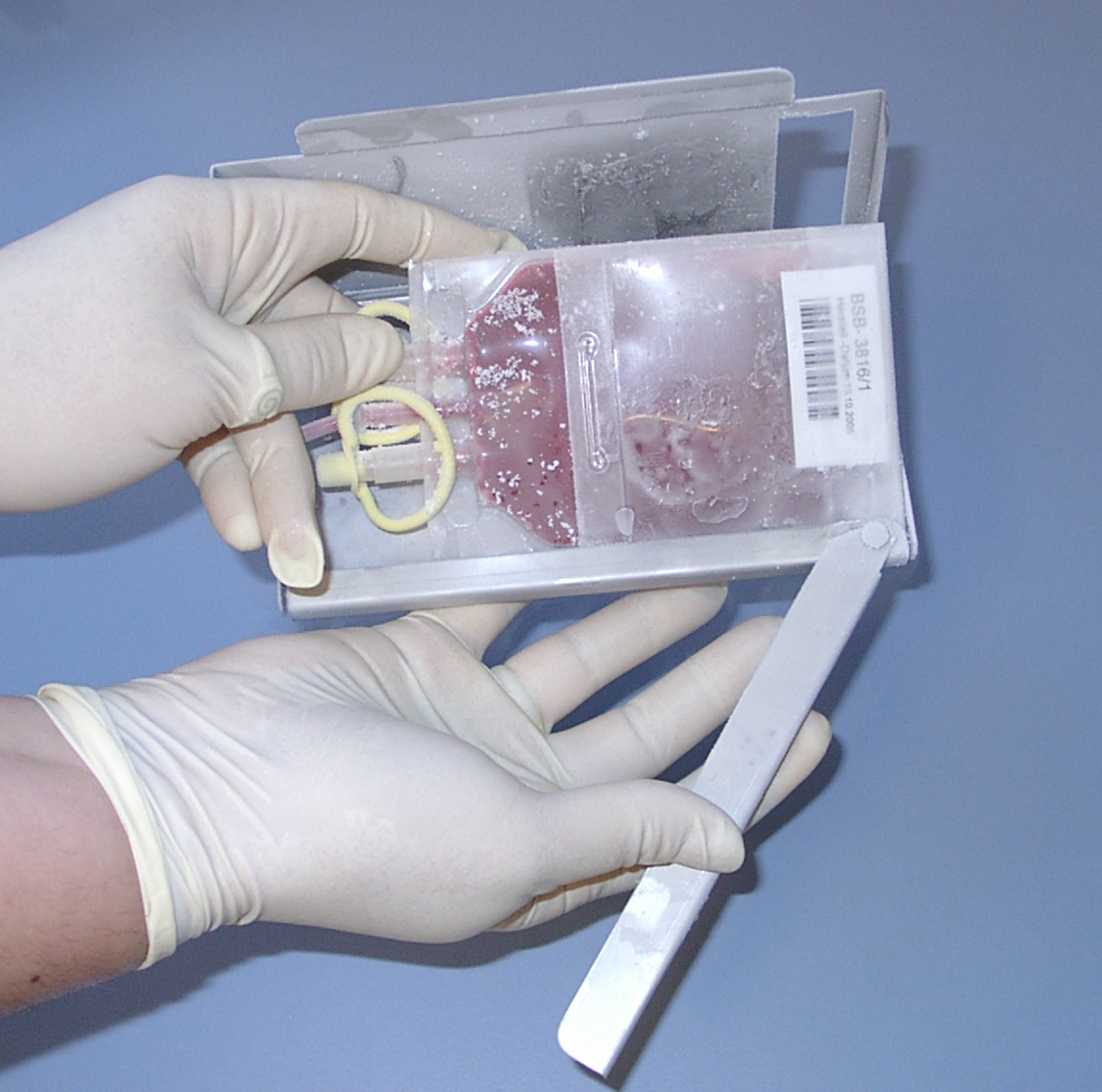
14. Pictures of cord blood units in the inventory
15. Infectious Disease Marker (IDM) CURRENTLY performed.
Holiday Calendar
- EDIT THE CALENDAR
Customise the different types of events you'd like to manage in this calendar.
#legIndex/#totalLegs - RESTRICT THE CALENDAR
Optionally, restrict who can view or add events to the team calendar.
#legIndex/#totalLegs - SHARE WITH YOUR TEAM
Grab the calendar's URL and email it to your team, or paste it on a page to embed the calendar.
#legIndex/#totalLegs - ADD AN EVENT
The calendar is ready to go! Click any day on the calendar to add an event or use the Add event button.
#legIndex/#totalLegs - SUBSCRIBE
Subscribe to calendars using your favourite calendar client.
#legIndex/#totalLegs